BioDiem has an existing revenue-generating business from the licensing of its Live Attenuated Influenza Virus (LAIV) vaccine technology to partners in India and China. However, BioDiem is also focused on the development of new vaccines and therapies. BioDiem’s in-house technologies, and its expertise surrounding the LAIV means the potential exists to design new vaccines, and use the LAIV as a ‘shell’ or ‘vector’ to jump-start the immune system and treat or prevent disease.
LAIV Vector
Viruses have the ability to generate proteins prolifically. Viral vectors are viruses which are used to deliver antigens i.e. specific disease-proteins to generate a desirable immune response in the person who has been vaccinated. A wide range of diseases can be targeted depending on the choice of antigen which is presented by the viral vector. Various viruses have been tried for this purpose i.e. to produce specific proteins of diseases which are unrelated to the carrier virus. Unfortunately these viruses often have disadvantages. For example, they are weak and therefore by being weak, are safe, but often may be too weak to generate the strong immune response which is needed for protection or treatment.
The LAIV vector research aims to produce a platform technology to assist in the design of vaccines for different diseases including cancers. The advantage of the LAIV virus technology is that it has a documented safety profile from its use in Russia over many years and through its use in the European clinical trial program as an influenza vaccine.
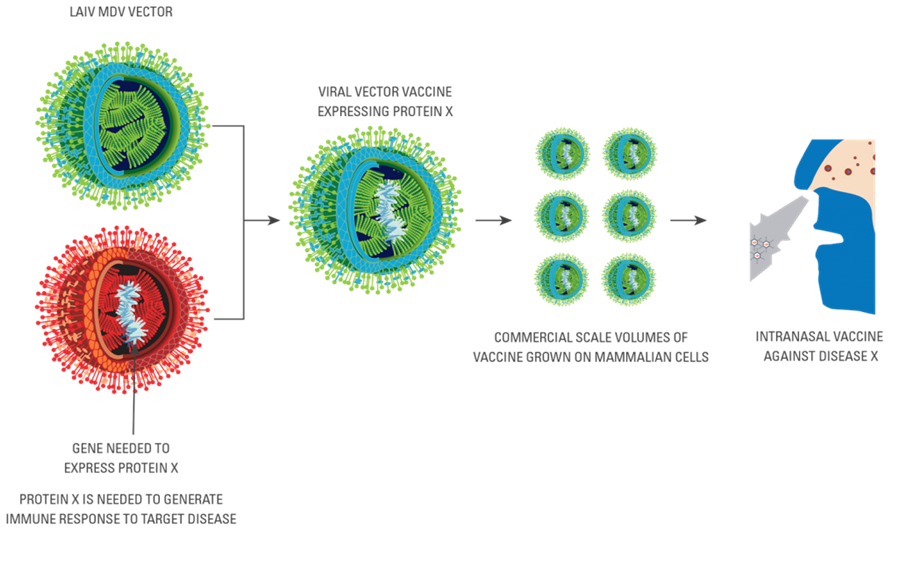
BioDiem is seeking interest from parties with expertise to collaborate in the next stages of the program leading to proof-of-concept. For further information, please contact info@biodiem.com.